Residency Program - Case of the Month
August 2011 - Presented by Melissa Rodgers-Ohlau, M.D.
Answer:
Uterine Endometrioid Adenocarcinoma, pT1aNoMx, with synchronous Ovarian Endometrioid Adenocarcinoma, pT1aNoMx, in the setting of Lynch Syndrome
Histological description
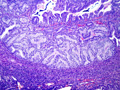
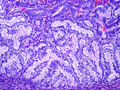
Microscopic examination of the uterus reveals a mass consisting of back to back glands with no intervening stroma and occasional papillary formations (villoglandular pattern) [Figure 5]; scattered glands contain necrotic or mucinous debris; the cells lining the glands have a moderate amount of eosinophilic to amphophilic cytoplasm and ill-defined cell borders at the luminal surface; the nuclei are uniform, oval, with fine chromatin and small nucleoli, no mitotic figures; several small foci of superficial myometrial invasion are identified (1 mm in depth) (Figure 6]; no lymphovascular invasion is seen; foci of complex hyperplasia with atypia are also present [Figures 7 & 8]; the cervix, serosa and left fallopian tube are uninvolved by malignancy.
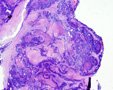
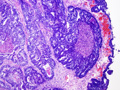
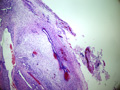
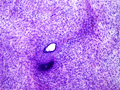
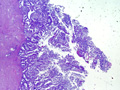
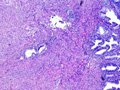
Microscopic examination of the left ovary reveals an intraparenchymal mass (no surface involvement) consisting of back to back glands with cribriform bridging and intraluminal necrotic debris [Figures 1 & 2]; uniform nuclei with little atypia; no mitiotic figures; no lymphovascular invasion; and rare foci of endometriosis [Figures 3 & 4].
Microscopic examination of the remainder of the specimens, including the right ovary, right fallopian tube and specimens obtained for staging purposes were all histologically unremarkable, negative for malignancy.
Discussion:
We initially staged the uterine adenocarcinoma as pT3a, believing that the tumor had metastasized to the ovary (per the CAP protocol for endometrial carcinoma, tumor involvement of the adnexa, either by direct extension or metastasis is staged as such). However, after further consideration, we amended our initial diagnosis to "uterine endometrioid adenocarcinoma, pT1aNoMx, with synchronous ovarian endometrioid adenocarcinoma, pT1aNoMx," based on an extensive list of criteria set forth by Scully et al. When assessing synchronous tumors of the endometrium and ovary, one needs to determine whether the malignancy arose in the uterus and extended or metastasized to the ovary; whether the malignancy arose in the ovary and extended or metastasized to the uterus; or whether the malignancies represent two synchronous primary tumors. Scully et al. established separate sets of criteria for each situation in order to facilitate the decision-making process. Using the criteria for Endometrial Primary, Ovarian Secondary (Table 1), we ruled out an endometrial primary based on the fact that: there was no significant difference in the relative sizes of the endometrial and ovarian tumors; there was no deep myometrial invasion with either direct adnexal extension or lymphovascular invasion; the ovarian tumor was neither bilateral nor multinodular; the ovarian tumor was not located in the hilum, nor were there any surface implants or lymphovascular invasion; and ovarian endometriosis was present, diminishing support for metastasis to the ovary. Based on criteria for Ovarian primary, endometrial secondary (Table 2), we ruled out an ovarian primary with metastasis to the endometrium because: there was no direct extension from the ovary either into the outer wall of the uterus or into the fallopian tube; again, there was no significant difference in the relative sizes of the ovarian and endometrial tumors; and there was atypical hyperplasia in the endometrium, diminishing support for ovarian metastasis to the endometrium. Our final decision was solidified based on criteria for Endometrioid tumors of the endometrium and ovary; Independent primary tumors (Table 3) as follows: there was only superficial myometrial invasion of the endometrial tumor; again, there was no lymphovascular invasion in either the endometrial or ovarian tumor; there was no evidence of tumor spread from either the endometrium or the ovary; the ovarian tumor was unilateral and intraparenchymal; atypical endometrial hyperplasia was present, supporting development of a primary endometrioid adenocarcinoma of the uterus; and there was endometriosis in the ovary, supporting development of a primary ovarian endometrioid adenocarcinoma.
These findings, synchronous primary endometrial and ovarian tumors, are not surprising in the setting of Lynch syndrome. The patient had been diagnosed 9 years earlier, at the age of 30, with an ileocecal valve mass (pT3N0M0, poorly differentiated adenocarcinoma with mucinous features) for which she received a right hemicolectomy. Due to her young age at initial diagnosis, Lynch syndrome was suspected and microsatellite instability (MSI) testing was performed. Eight of nine markers were found to be unstable; subsequent germline testing was performed and was positive for variant mutation in MLH-1.
Lynch syndrome, also known as hereditary nonpolyposis colorectal cancer syndrome (HNPCC), was named for Dr. Henry Lynch who characterized the syndrome in the 1960's. He coined the term HNPCC but it is more of a misnomer as individuals who inherit one of the genetic mutations involved in the syndrome are susceptible to a variety of cancers, not just colon cancer. Lynch syndrome is an autosomal dominant disorder caused by a germline mutation in one of several DNA mismatch repair (MMR) genes. The syndrome is typified by early age-of-onset and multiple cancers. There is a markedly increased risk of colorectal cancer (approximately 70%) as well as cancer of the endometrium, ovary, upper genitourinary tract, stomach, small bowel, pancreaticobiliary tract and brain. The risk of developing these cancers is influenced by the regional prevalence of the cancer. For example, gastric cancer is more common in Japan, Korea and China, therefore, the risk of developing gastric cancer in Lynch syndrome would be higher in one of these countries than in the U.S. The spectrum of cancers to which an individual may be susceptible and the age of onset can also vary based on the MMR mutation inherited. For example, the risk of developing endometrial cancer, the most common extracolonic cancer in Lynch syndrome, ranges from 27-71%, based on whether the individual inherits mutations in MLH1 (27%), MSH2 (40%) or MSH6 (71%).
During DNA replication and base pairing, MMR genes/protein products function to correct base substitution mismatches and insertion-deletion mismatches. The most commonly mutated MMR genes in Lynch syndrome are MLH1, MSH2, MSH6 and PMS2. In Lynch syndrome, defects in the aforementioned MMR genes are inherited as germline mutations (all cells in that individual have an inherently mutated allele). The second allele is then knocked out in some fashion, for example through mutation or epigenetic silencing (methylation). With the MMR system nonfunctional, numerous key genes – such as those that control cell growth or control apoptosis - acquire mutations, leading to oncogenesis. Microsatellite instability (MSI) is also a direct result of mismatch repair dysfunction. Microsatellites are regions of repetitive nucleotide sequences; when MMR genes are nonfunctional, these microsatellite regions are more prone to expansion and contraction. Using PCR and a panel of standard DNA probes for these microsatellite regions, microsatellite instability testing (on either colonic or endometrial tumors) is one of the initial means of assessing MMR dysfunction. If 30% or more of the MSI markers show expansion or contraction within the microsatellite regions of the patient's tumor, compared with normal tissue from the patient, the tumor is reported to have "High Level of Microsatellite Instability (MSI-H)." More than 90% of tumor tissue from patients with Lynch syndrome shows MSI-H. While the test is very sensitive, it is not specific for Lynch syndrome because approximately 15% of sporadic colorectal cancer is positive for MSI-H (usually due to MMR silencing by hypermethylation of the promoter region of MLH1). To differentiate between sporadic MSI-H and MSI-H associated with Lynch syndrome, either the methylation status of the MLH1 promoter may be assessed or testing for V600E activating mutations in BRAF may be performed. With regard to the latter, if the V600E mutation is present, Lynch syndrome may be ruled out; the reason for this is currently unknown.
Because MSI testing lacks specificity, immunohistochemical (IHC) staining for the most commonly mutated genes/proteins should also be performed (either on colonic or endometrial tumors). In general, positive staining indicates that the MMR protein product is functional while negative staining indicates an MMR mutation. In some instances a truncated protein may be mutated in such a way that the antigenic site (for IHC) is still intact, giving a false positive result (in other words, the presence of the MMR mutation is masked). As a result, if IHC staining is positive but the index of suspicion for Lynch syndrome is high, it is important to pursue further testing. Definitive diagnosis of Lynch syndrome is based on germline testing. Commercial testing is available for MSH2 and MLH1; on special request, testing for MSH6 and PMS2 can also be performed.
Table 1*
|
Endometrial primary, ovarian secondary |
|
1. Histologic similarity of the tumors. |
|
2. Large endometrial tumor, small ovarian tumor (s). |
|
3. Atypical endometrial hyperplasia additionally present. |
|
4. Deep myometrial invasion: a. Deep extension into the adnexa. b. Vascular space invasion in myometrium. |
|
5. Spread elsewhere in typical pattern of endometrial carcinoma. |
|
6. Ovarian tumor bilateral and/or multinodular. |
|
7. Hilar location, vascular space invasion, surface implants or combination in ovary. |
|
8. Ovarian endometriosis absent. |
|
9. Aneuploidy with similar DNA indices or diploidy of both tumors. # |
|
10. Similar molecular genetic or karyotypic abnormalities in both tumors. |
Table 2*
|
Ovarian primary, endometrial secondary |
|
1. Histologic similarity of tumors. |
|
2. Large ovarian tumor, small endometrial tumor. |
|
3. Ovarian endometriosis present. |
|
4. Location in ovarian parenchyma. |
|
5. Direct extension from ovary predominantly into outer wall of uterus. |
|
6. Spread elsewhere in typical pattern of ovarian carcinoma. |
|
7. Ovarian tumor unilateral (80-90% of cases) and forming single mass. |
|
8. No atypical hyperplasia in endometrium. |
|
9. Aneuploidy with similar DNA indices or diploidy of both tumors. # |
|
10. Similar molecular genetic or karyotypic abnormalities in both tumors. |
Table 3*
|
Endometrioid tumors of the endometrium and ovary: |
|
Independent primary tumors |
|
1. Histologic dissimilarity of the tumors. |
|
2. No or only superficial myometrial invasion of endometrial tumor. |
|
3. No vascular space invasion of endometrial tumor. |
|
4. Atypical endometrial hyperplasia additionally present. |
|
5. Absence of other spread of endometrial tumor. |
|
6. Ovarian tumor unilateral (80-90% of cases). |
|
7. Ovarian tumor located in the parenchyma. |
|
8. No vascular space invasion, surface implants or predominant hilar location in ovary. |
|
9. Absence of other evidence of spread of ovarian tumor. |
|
10. Ovarian endometriosis present. |
|
11. Different ploidy of DNA indices, if aneuploid, of the tumors. # |
|
12. Dissimilar molecular genetic or karyotypic abnormalities in the tumor. |
* All tables from Scully et al.
# The possibility of tumor heterogeneity must be taken into account in the evaluation of ploidy findings.
Microscopic photographs:
|
Ovary |
|||
 |
 |
 |
 |
|
Uterus |
|||
 |
 |
 |
 |
References:
Ahnen, Dennis J, M.D. and Axell, Lisen, M.S. "Clinical features and diagnosis of Lynch Syndrome (hereditary nonpolyposis colorectal cancer)." Up To Date. January 2011.
CAP Cancer Protocols. Colon and Rectum.
http://www.cap.org/apps/cap.portal?_nfpb=true&cntvwrPtlt_actionOverride=%2Fportlets%2FcontentViewer%2Fshow&_windowLabel=cntvwrPtlt&cntvwrPtlt%7BactionForm.contentReference%7D=committees%2Fcancer%2Fcancer_protocols%2Fprotocols_index.html&_state=maximized&_pageLabel=cntvwr.
CAP Cancer Protocols, Ovary.
http://www.cap.org/apps/cap.portal?_nfpb=true&cntvwrPtlt_actionOverride=%2Fportlets%2FcontentViewer%2Fshow&_windowLabel=cntvwrPtlt&cntvwrPtlt%7BactionForm.contentReference%7D=committees%2Fcancer%2Fcancer_protocols%2Fprotocols_index.html&_state=maximized&_pageLabel=cntvwr.
CAP Cancer Protocols, Endometrium.
http://www.cap.org/apps/cap.portal?_nfpb=true&cntvwrPtlt_actionOverride=%2Fportlets%2FcontentViewer%2Fshow&_windowLabel=cntvwrPtlt&cntvwrPtlt%7BactionForm.contentReference%7D=committees%2Fcancer%2Fcancer_protocols%2Fprotocols_index.html&_state=maximized&_pageLabel=cntvwr.
Poynter, J.N., et al. "Molecular characterization of MSI-H colorectal cancer by MLHI promoter methylation, immunohistochemistry, and mismatch repair germline mutation screening." Cancer Epidemiol Biomarkers Prev. 2008 Nov;17(11):3208-15
Scully et al. "Tumors of the ovary, maldeveloped gonads, fallopian tube and broad ligament." Atlas of Tumor Pathology. Bethesda, MD: Armed Forces Institute of Pathology; 1998.
Soliman, Pamela, et al. "Synchronous primary cancers of the endometrium and ovary: a single institution review of 84 cases." Gynecologic Oncology. 94 (2004) 456-462.

 Meet our Residency Program Director
Meet our Residency Program Director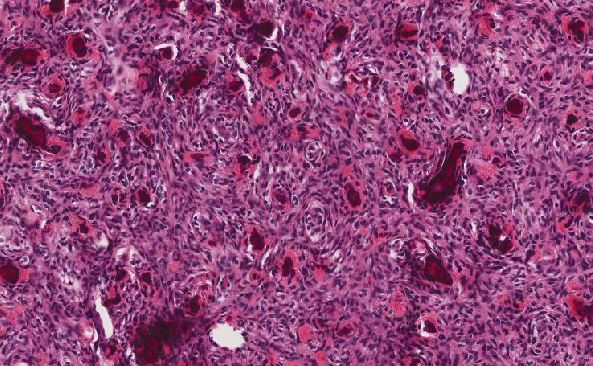
 LeShelle May
LeShelle May Chancellor Gary May
Chancellor Gary May