Bits & pieces
Liquid biopsy could ease the way to immunotherapy for lung cancer
A collaboration of researchers at UC Davis, Genentech and Foundation Medicine are the first to show that a blood-based test to assess tumor mutational burden (TMB) accurately identifies non-small cell lung cancer patients who could benefit from immunotherapies called checkpoint inhibitors. The blood test offers a much less invasive and more repeatable alternative to tissue testing. The study was published in August in Nature Medicine.
“We wanted to know if we could transfer this TMB assay from tissue to blood,” said David Gandara, who directs the cancer center’s Thoracic Oncology Program and is first author on the paper. “We succeeded, establishing a TMB level in blood that correlates well with similar levels in tissue and was associated with favorable patient outcomes.”
Patients with higher TMB are often better candidates for checkpoint inhibitors.
“There are patients for whom the biopsy is inadequate from the start, or the tissue is used for routine pathology and we don’t have enough tissue left to do either genomic testing or tissue TMB,” Gandara said. “If we can do it in blood in one test, that offers many advantages for patients who have had an inadequate biopsy.”
Gandara said the test has already received Breakthrough Designation, the first step towards full FDA approval.
Necessity breeds invention for four-time cancer survivor
Cinde Dolphin had nine surgeries in 10 years for lung and breast cancer, but the four-time cancer survivor turned pain and frustration into an opportunity. The device she created to make patients like her more comfortable after surgery has since been embraced and endorsed by medical professionals at UC Davis and beyond.

“It’s been part of her healing process,” said Scott Christensen, a medical oncologist at the cancer center. “Doing something positive for others is very empowering.”
Like many post-surgery patients, Dolphin required a drain implanted near the incision site to prevent fluid build-up in her body. About a foot of thin plastic tubing carried the fluid to a plastic bulb that had to be emptied when full.
The drain can be cumbersome, uncomfortable, and get in the way of daily life. Dolphin wore them for weeks at a time.
Dolphin’s plastic surgeon recommended she use an apron from Home Depot to carry the drains, which hang awkwardly from the body. She thought there must be a better way.
“The apron pockets effectively kept the drain suspended, but the canvas construction prevented me from showering with it,” Dolphin said.
Dolphin took matters into her own hands. She went to a Dollar Store, loaded her cart with mesh laundry bags and, with help from family members, created a prototype.
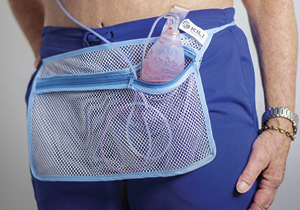
“My brilliant nephew was willing to serve as the company’s seamstress,” she said. “I re-engineered the bags by sewing ¾-inch white bias tape to the top portion above the zipper. The design was ideal for 24/7 use, even when bathing. I knew I was on to something.”
Christensen gave Dolphin feedback on the product and connected her to other medical staff for their opinions.
“I thought it was a great idea,” recalled Gary Kurtz, manager of the Post-Anesthesia Care Unit (PACU). “A patient saw a problem and wanted to help others.”
Kurtz placed an order for the UC Davis PACU and gave the carriers to his post-surgery patients. Nurses and patients alike responded positively. Today, each patient who needs a drain gets Dolphin’s Kili Medical Drain Carrier. The discrete, inexpensive pouch is lightweight, can be worn in the shower and allows patients more mobility.
The name “Kili” comes from Dolphin’s volunteer trip as a business advisor to women in a micro-loan cooperative near Mt. Kilimanjaro in Tanzania. She visited the village after she recovered from her final cancer surgery and paid homage to the families she met by naming the device for them.
“The experience had a profound impact on my life,” Dolphin said. “Locals call Kilimanjaro ‘Kili,’ and it seemed a fitting way to honor the ‘mamas’ I met.”
With a clean bill of health and a new business venture, Dolphin sells the Kili Medical Drain Carrier for $14.99 in her retail shop online and directly to several Sacramento-area hospitals. She hopes to one day offer a similar solution for patients who wear colostomy bags.
“Thanks to everyone — including the UC Davis medical team and my network of friends and business associates — I am reminded that everything can be used for good, even after multiple cancer diagnoses.”
Saturday adult infusions

On Oct. 6, the North Building of the Cancer Center opened to adult oncology and non-oncology infusion patients. This new option provides an additional resource and more flexibility to patients who prefer Saturday appointments. To start, 12 chairs will be available from 8 a.m. to 6 p.m., and the number will grow as demand increases.
“Opening for six days a week allows us to meet the needs of our patients who can now avoid unnecessary hospital admissions or trips to emergency rooms for chemotherapy, blood transfusions or hydration on the weekend,” said Kristin Mensonides, director of oncology services. “We look forward to welcoming patients with open arms!”
Specialty clinic for sarcoma patients opens
The UC Davis Comprehensive Cancer Center has opened a multidisciplinary clinic specializing in pediatric, adolescent and young adult sarcoma.
The sarcoma clinic, located in the Pediatric Oncology Clinic at the Cancer Center, is the only one of its kind in the Sacramento region. The treatment team sees patients the first and third Thursday of each month. Patients have access to a full range of experts, including specialists in pediatric and medical oncology, sarcoma surgery, nursing case management and others under one roof.
The clinic allows for seamless care across specialties to assure the best possible outcomes for patients with bone and soft tissue sarcoma. Patients also have onsite access to specially trained physical therapists, social workers, nutritionists and other supportive care professionals to facilitate healing and survivorship.
“Advances in cancer have been the result of a multidisciplinary approach,” said Marcio Malogolowkin, chief of pediatric oncology at the cancer center. “Bringing experts together in the same room with the same patient increases our ability to communicate, improve our understanding of patients’ needs and deliver better care.”
Bone and soft tissue sarcomas are a very rare group of connective tissue malignancies, and successful treatment outcomes require a collaborative, multidisciplinary approach, which may include surgery, chemotherapy or radiation therapy. The clinic also offers various clinical trials of new treatment approaches.
The lead treatment team includes medical oncologists Elysia Alvarez and Marcio Malogolowkin, orthopedic surgical oncologists Steven Thorpe and Lor Randall, surgery nurse practitioner Abigail Inkster, social worker Robyn Peace, nurse care coordinators Yvonne Roach and Kristine Ahlberg and physical therapist Jennifer Beane. The team also includes additional experts in surgery, radiation oncology, radiology and pathology.
Addressing vast cancer disparities among Latinos, Asians and Blacks:
Davis leads precision medicine study to improve outcomes
UC Davis Comprehensive Cancer Center in September received a $6.3 million grant from the National Cancer Institute’s (NCI) Center to Reduce Cancer Health Disparities for a 5-year study to tease out why some ethnic and racial minority groups fare worse than whites when they get cancer and to find more precise treatments to improve their chances of survival.

The collaborative study engages four of the University of California’s NCI-designated comprehensive cancer centers: UC Davis Comprehensive Cancer Center, Jonsson Comprehensive Cancer Center at UCLA, UCSF Helen Diller Family Comprehensive Cancer Center and the Chao Family Comprehensive Cancer Center at UC Irvine.
Under the grant, entitled “University of California Minority Patient-Derived Xenograft (PDX) Development and Trial Center” or UCaMP, tumor samples from patients will be delivered to UC Davis for the research, most of them from minority patients and the rest from non-Hispanic whites for use as controls. The samples will be used to grow tumors (or xenografts) in mice. They are then replicated so that each can be used to test a different drug. The goal is to generate at least 200 PDXs for use in two research projects.
The study will focus on four types of cancer that disproportionately affect minorities: lung, liver, gastric (stomach) and bladder. The first project in the study will focus on gastric and liver cancer among Latinos and Asian Americans, Pacific Islanders and Native Hawaiians, and the second will examine lung and bladder cancers among African Americans. Researchers will use PDXs to discern their genetic characteristics and test potential targeted drugs to treat them.
When compared with non-Hispanic whites, African Americans have lower survival from lung and bladder cancer, even when diagnosed at the same stage of the disease.
“We are trying to address why this happens,” said Chong-xian Pan, a medical oncologist and study principal investigator who leads that project. “Socio-economics could be a factor, but we also want to see if there are biological factors contributing to this disparity. PDXs are a great model, not only to study the biological differences of cancers from non-Hispanic whites and African Americans, but also to facilitate cancer drug development and precision medicine.”
Luís Carvajal-Carmona, a UC Davis cancer genomicist, expert in the genetics of Latinos and a principal investigator, will lead the stomach and liver cancer project. He said the study is an effort to correct a major discrepancy in existing cancer genomics research to date — the lack of minority patient representation, which is essential for the development of targeted, or personalized, treatments.
“Minority patients have the worst cancer outcomes for multiple reasons,” he said. “One of them is that they come in with more advanced tumors because many don’t have proper access to health care. But also because there are not enough data and models for the scientific community to develop precision medicine studies in them. So we want this NCI-funded research to be a magnet for minority-focused clinical trials.”
Moon Chen, a UC Davis population scientist, expert in Asian cancer health disparities and a principal investigator, said PDXs have advantages over other research tools.
“They contribute to a better understanding of the underlying mechanisms of resistance and elucidate potential biological determinants of treatment responses and cancer health disparities for these tumor sites,” he said.
UC Davis research informs new national cervical cancer screening recommendation

Joy Melnikow, director of the UC Davis Center for Healthcare Policy and Research, led a comprehensive analysis of eight clinical trials and four cohort studies on cervical cancer screening. The study results show that while Pap smears are still highly effective for detecting pre-cancerous cells and cancer, testing for the virus that causes these cancers also is an excellent screening tool.
The findings, published in the Journal of the American Medical Association (JAMA) in August, were used to inform the U.S. Preventive Services Task Force, which published its updated recommendations on cervical cancer screening in the same issue of the journal.
The task force now recommends screening for cervical cancer every three years with Pap smear alone in women aged 21 to 29 years. For women aged 30 to 65 years the task force recommends screening every three years with a Pap smear alone, every five years with human papilloma virus (HPV) testing alone, or every five years with HPV testing in combination with a Pap smear.
It’s never too late to stop: Healing and health benefits start as soon as 20 minutes after you quit smoking
- 20 minutes: Blood pressure and pulse return to normal

- 8-12 hours:Blood oxygen and carbon monoxide levels return to normal

- 48 hours:ability to taste and smell is greatly improved

- 72 hours:breathing becomes easier and energy levels increase

- 2-12 weeks:circulation improves and lung function increases
- 12 months:excess risk of coronary heart disease is reduced by about half and declines gradually thereafter



