Case of the Month
June 2016 - Presented by Dr. Saba Ali & Dr. Mingyi Chen
Answer:
B.) BRAF
Histological Description
Brain Frontal Mass:
H&E sections of the excisional biopsy show sheets of neoplastic epithelioid cells. These cells have round nuclei with smooth to slightly irregular nuclear borders and variably prominent red nucleoli. The eosinophilic cytoplasm is moderate. The cells stain strongly positive for S100, HMB45, and MelanA on immunohistochemistry and are negative for lymphocytic markers.
Peripheral blood smear, bone marrow biopsy, and flow cytometry:
Sections of the bone marrow biopsy shows a hypocellular marrow (20-30% of cellularity) with an atypical B-cell interstitial infiltrate. The cells are small to medium in size with smooth nuclear borders, clumped chromatin with variable prominent nucleoli and are rimmed by pale-gray cytoplasm. Circulating in the peripheral blood is an atypical lymphocyte with extensive radial cytoplasmic processes projecting from the surface. The differential diagnosis given the morphologic features of the peripheral blood and bone marrow includes but is not limited to HCL, splenic marginal zone lymphoma, and hairy cell leukemia variant.
Flow cytometry of the bone marrow demonstrates an abnormal population of kappa restricted monotypic mature B-lymphocytes that express CD19+, CD20 (bright), CD11c+, CD103 (dim), CD25 (dim).
Immunohistochemical staining for Annexin A1 in these cells is equivocal due to positive staining in the myeloid precursors. Immunohistochemical stain for DBA-44 was positive.
Discussion:
(B)RAF (Rapidly Accelerated Fibrosarcoma) gene mutations are reportedly associated with papillary thyroid carcinoma (30-70%)(5), malignant melanoma (50%)(1), HCL, adenocarcinoma of the lung and colon (5-20%)(1) and ovarian cancer (15-30%)(1). A majority of HCL cases are associated with BRAF mutations, with only a few cases due to mutation of IGHN @ gene instead(1) and the more recently reported CDKN1B gene(7). Additionally, BRAF mutations have a higher association with metastatic melanomas with an occult primary site, and are reportedly more aggressive in addition to occurring in areas that are not exposed to sunlight(3). The association of HCL with concomitant melanoma has previously been reported(4), however more recent studies have shown there is no significant correlation in HCL and malignant melanoma occurrence despite the common molecular pathogenesis. In a study cohort at Sloan Kettering Memorial Center, 267 cases of HCL were examined for incidence of concomitant melanoma and non-melanoma skin cancer. Eleven cases (4.4%) were associated with melanoma and twenty one cases (6.9%) were associated with nonmelanoma skin cancer (squamous cell carcinoma and basal cell carcinoma). Molecular analysis performed on nine of these cases (two melanoma cases) showed a RAS mutation in 33.3%. BRAF mutations were not identified in any of the secondary skin samples(7).
The proto-oncogenes of the RAF family include ARAF, BRAF, and CRAF. BRAF (serine/threonine-protein kinase B-Raf) was first isolated in 1993 from a rodent retrovirus and its malignant potential was discovered when inoculated normal mouse fibroblasts transformed into neoplastic fibrosarcoma cells.(6) In 2002, BRAF was first found in human tumors(2). Normally, dividing cells display surface receptors that bind to growth factors present in the surrounding environment. This leads to activation of a family of gene products that stimulate cell growth (RET). Activated RET binds to RAF by a GTP-dependent mechanism. Subsequent phosphorylation of serine and threonine residues by RAF present on the proteins of the mitogen—activated protein (MAP) kinase pathway (MEK/ERK) send signals to the nucleus. Mutated RAF leads to constitutive activation of the downstream signaling pathways. This allows the cell to bypass the G1 restriction point of the cell cycle with upregulation of cyclin D1, resulting in unchecked cellular proliferation and survival.(5)
Somatic mutations of the BRAF gene located on chromosome 7q34 most commonly occur at codon 600 (V600e) in the activation loop and are characterized by a substitution of the amino acid valine to glutamic acid or lysine(1). Two pharmaceutical agents currently approved by the FDA for therapy in BRAF mutated cancers include Vemurafenib (2011, Plexxikon and Genentech) and combined therapy with Dabrafenib/Trametinib. Resistance can occur, and plausible mechanisms include receptor tyrosine kinase such as the platelet derived growth factor receptor and upregulation of upstream pathways(5). In regard to the occurrence of melanoma in patients with HCL, it has been hypothesized the treatment with BRAF inhibitors in these patients results in secondary activating mutations of the RAS proto-oncogenes, particularly KRAS, and thus the initial therapy itself may play a role in the concomitant incidence of skin cancer(5). Further studies regarding this association and associated underlying mechanisms are necessary.
References
- Blachly JS, Lozanski G, Lucas DM, Grever MR, Kendra K, Andritsos LA. Cotreatment of hairy cell leukemia and melanoma with the BRAF inhibitor dabrafenib. Journal of the National Comprehensive Cancer Network. 2015;13(1):9-13.
- Helen Davies et al. Mutations of the BRAF gene in human cancer. Nature. 27 June 2002; 417, 949-954.
- Zepeda-Lopez PD, Salas-Alanis JC, Toussaint-Caire S, Gutierrez-Mendoza D, Vega-Memije E, Silva SL, Fajardo-Ramírez OR, Alcazar G, Moreno-Treviño MG, Saldaña HA. BRAF Mutation (V600E) Prevalence in Mexican Patients Diagnosed with Melanoma. Case Reports Oncology. 2016 Apr 21;9(1):241
- Toner et al. Lethal skin cancers in hairy cell leukemia. European Journal of Haemoatology. 1987; 39:82-85
- Su et al. Resistance to Selective BRAF Inhibition Can Be Mediated by Modest Upstream Pathway Activation. Cancer Res. 2012; 72(4); 969–78.
- Huang T, Karsy M, Zhuge J, Zhong M, Liu D. B-Raf and the inhibitors: from bench to bedside. Journal of Hematology & Oncology. 2013;6:30.
- Watts et al. Melanoma and non-melanoma skin cancers in hairy cell leukaemia: a Surveillance, Epidemiology and End Results population analysis and the 30-year experience at Memorial Sloan Kettering Cancer Center. British Journal of Haematology. 2015; 171 (1):84–90.
- Robak T, Smolewski P. New mutation in hairy cell leukemia. Blood Journal. 20 August 2015; 126 (8):930 – 931.

 Meet our Residency Program Director
Meet our Residency Program Director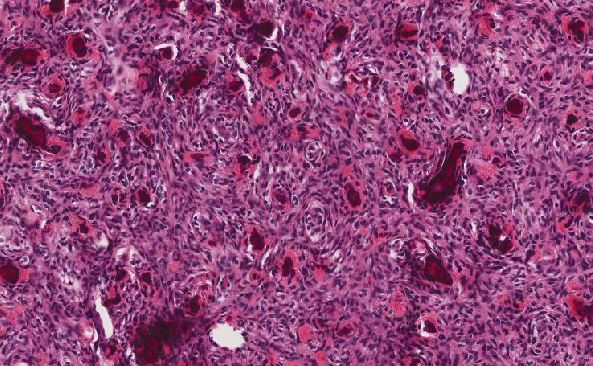
 LeShelle May
LeShelle May Chancellor Gary May
Chancellor Gary May