Residency Program - Case of the Month
May 2015 - Presented by Dr. Amir Ghorbani & Dr. Mirna Lechpammer
Answer:
Skeletal muscle with myopathic features consistent with CENTRONUCLEAR MYOPATHY.
Centronuclear Myopathies
Centronuclear myopathies (CNM) are a group of congenital myopathies where cell nuclei are abnormally located in skeletal muscle cells. In CNM the nuclei are located at a position in the center of the cell, instead of their normal location at the periphery.
History:
In 1966, Dr. Spiro (a New York City neurologist) published a medical report of a boy with myopathy, which upon muscle biopsy, showed that the nuclei of the muscle cells were located in the center of the muscle cells, instead of their normal location of the periphery¹. The nuclear appearance reminded him of the nucleus-in-the-center appearance during the “myotubular” stage of embryonic development. Thus, he coined the term "myotubular myopathy". Spiro speculated that the embryonic muscle development he had seen in the boy was due to growth arrest during the myotubular phase, causing the myopathy.
More than three decades later, it is not fully understood whether this theory regarding halted (or delayed) embryonic muscle development is correct. Some research suggests that this theory may be acceptable for infant-onset myotubular myopathy (mutations at the MTM1 gene on the X chromosome), but may not be acceptable for the autosomal forms of centronuclear myopathy.²
Epidemiology:
The overall incidence of myotubular myopathy is 1 in 50,000 male live births. The incidence of other centronuclear myopathies is extremely rare, with there only being nineteen families identified with CNM throughout the world.³
Clinical Presentations
As with other myopathies, the clinical manifestations of MTM/CNM are most notably muscle weakness and associated disabilities. Congenital forms often present with neonatal low muscle tone, severe weakness, delayed developmental milestones (particularly gross motor milestones such as head control, crawling, and walking) and pulmonary complications (presumably due to weakness of the muscles responsible for respiration). While some patients with centronuclear myopathies remain ambulatory throughout their adult life, others may never crawl or walk and may require wheelchair use for mobility. There is substantial variability in the degree of functional impairment among the various centronuclear myopathies.
X-linked myotubular myopathy was traditionally a fatal condition of infancy, with life expectancy of usually less than two years. There appears to be substantial variability in the clinical severity for different genetic abnormalities at that same MTM1 gene. Further, published cases show significant differences in clinical severity among relatives with the same genetic abnormality at the MTM1 gene. Most truncating mutations of MTM1 cause a severe and early lethal phenotype, while some missense mutations are associated with milder forms and prolonged survival (up to 54 years).4
Centronuclear myopathies typically have a milder presentation and a better prognosis. Recently, researchers discovered mutations at the gene dynamin 2 (DNM2 on chromosome 19, at site 19p13.2), responsible for the autosomal dominant form of centronuclear myopathy.5 This condition is now known as dynamin 2 centronuclear myopathy (abbreviated DNM2-CNM). Research has indicated that patients with DNM2-CNM have a slowly progressive muscular weakness usually beginning in adolescence or early adulthood, with an age range of 12 to 74 years.
Diagnosis:
Creatine kinase (CK) usually is in the normal range, although occasionally it is elevated. Myopathic changes may be seen on the electromyography (EMG). There is substantial overlap between the electrodiagnostic findings the various types of myopathy. Thus, electrodiagnostic testing can help distinguish neuropathy from myopathy, but is not effective at distinguishing which specific myopathy is present, here muscle biopsy and perhaps subsequent genetic testing are required.
There are three types of CNM:
-
X-linked Recessive: The X-linked form of MTM/CNM is the most commonly diagnosed type. Almost all cases of X-linked MTM occurs in males.
Male infants have marked hypotonia and skeletal muscle weakness. Respiratory muscle impairment leads to respiratory failure. Facial weakness, ptosis, and extraocular muscle weakness are common, and impaired bulbar function contributes to feeding difficulty. Rarely, abnormal genital development (ambiguous genitalia or severe hypospadias) occurs.6 Heterozygous female carriers of the associated mutations may present with limb girdle and facial weakness.7
Prenatal history in X-linked myotubular myopathy includes polyhydramnios (caused by impaired swallowing) and decreased fetal movement in 50 to 60 percent of cases.8 Many infants fail to establish effective breathing at birth.
Approximately one-third of severely affected patients die from respiratory complications during infancy and outcome is poor in many survivors. In one series of 55 males with the disorder, 64 percent survived longer than one year, although 80 percent were completely or partially ventilator-dependent.9 The disease appeared to be nonprogressive, and cognitive function was normal in the absence of significant hypoxic-ischemic injury. -
Autosomal recessive: Mutation in BIN1, RYR110 or TTN genes. relatively mild weakness and hypotonia that may be unrecognized in the neonatal period. Three subgroups have been described for autosomal recessive and sporadic presentations11:
• Early onset with ophthalmoparesis
• Early onset without ophthalmoparesis
• Late onset without ophthalmoparesis -
Autosomal dominant: Mutation in a gene called dynamin 2 (DNM2) on chromosome 19, and this particular condition is now referred to as dynamin 2 centronuclear myopathy (DNM2-CNM). Two of these are autosomal dominant12:
• A classic form characterized by late onset and slow progression
• A form similar to the classic but with diffuse muscle hypertrophy
Dynamin-2 Protein:
Dynamin-2 is a protein that in humans is encoded by the DNM2 gene Dynamins represent one of the subfamilies of GTP-binding proteins. Dynamins are associated with microtubules. Dynamins bind many proteins that bind actin and other cytoskeletal proteins. Dynamins can also self-assemble, a process that stimulates GTPase activity13. Mutations in this gene have been associated to cases of acute lymphoblastic leukemia14, or congenital myopathy (centronuclear type)15.
Missense mutations affecting the Middle domain of the dynamin 2 (DNM2) protein, a large GTPase implicated in membrane and cytoskeletal remodeling, have recently been demonstrated to cause the slowly progressive autosomal dominant centronuclear myopathy (DNM2-centronuclear myopathy)16. Moreover, DNM2 mutations restricted to the Pleckstrin Homology (PH) domain of the DNM2 gene have also been recently identified in patients with autosomal dominant intermediate Charcot-Marie-Tooth disease (DNM2-DI-CMTB), an hereditary motor and sensory axonal neuropathy17. More recently, a de novo mutation in the skeletal muscle ryanodine receptor (RYR1) gene has been demonstrated in 1 patient with centronuclear myopathy.
Treatment:
Currently there is no cure for myotubular or centronuclear myopathies. Treatment often focuses on trying to maximize functional abilities and minimize medical complications, and involvement by physicians specializing in Physical Medicine and Rehabilitation, and by physical therapists and occupational therapists.
Medical management generally involves efforts to prevent pulmonary complications, since lung infections can be fatal in patients lacking the muscle strength necessary to clear secretions via coughing. Medical devices to assist with coughing help patients maintain clear airways, avoiding mucous plugs and avoiding the need for tracheostomy tubes.
Monitoring for scoliosis is also important, since weakness of the trunk muscles can lead to deviations in spinal alignment, with resultant compromise of respiratory function. Many patients with congenital myopathies may eventually require surgical treatment of scoliosis.
References:
-
Spiro A, Shy G, Gonatas N (1966). "Myotubular myopathy. Persistence of fetal muscle in an adolescent boy". Arch Neurol 14 (1): 1–14. doi:10.1001/archneur.1966.00470070005001. PMID 4954227
-
Myotubular Myopathy, Autosomal Dominant Online Mendelian Inheritance in Man, OMIM. Johns Hopkins University, Baltimore, MD.
-
MTM1 analysis for Myotubular Myopathy The University of Chicago Genetic Services.
-
Laporte J, Biancalana V, Tanner S, Kress W, Schneider V, Wallgren-Pettersson C, Herger F, Buj-Bello A, Blondeau F, Liechti-Gallati S, Mandel J (2000). "MTM1 mutations in X-linked myotubular myopathy". Hum Mutat 15 (5): 393–409. doi:10.1002/(SICI)1098-1004(200005)15:5<393::AID-HUMU1>3.0.CO;2-R. PMID 10790201
-
Bitoun M, Maugenre S, Jeannet P, Lacène E, Ferrer X, Laforêt P, Martin J, Laporte J, Lochmüller H, Beggs A, Fardeau M, Eymard B, Romero N, Guicheney P (2005). "Mutations in dynamin 2 cause dominant centronuclear myopathy". Nat Genet 37 (11): 1207–1209. doi:10.1038/ng1657. PMID 16227997
-
Bartsch O, Kress W, Wagner A, Seemanova E. The novel contiguous gene syndrome of myotubular myopathy (MTM1), male hypogenitalism and deletion in Xq28:report of the first familial case. Cytogenet Cell Genet 1999; 85:310
-
Sutton IJ, Winer JB, Norman AN, et al. Limb girdle and facial weakness in female carriers of X-linked myotubular myopathy mutations. Neurology 2001; 57:900
-
Volpe JJ. Neuromuscular disorders: Muscle involvement and restricted disorders. In: Neurology of the Newborn, 5th ed, Saunders Elsevier, Philadelphia 2008. p.801
-
Herman GE, Finegold M, Zhao W, et al. Medical complications in long-term survivors with X-linked myotubular myopathy. J Pediatr 1999; 134:206.
-
Monnier N, Ferreiro A, Marty I, et al. A homozygous splicing mutation causing a depletion of skeletal muscle RYR1 is associated with multi-minicore disease congenital myopathy with ophthalmoplegia. Hum Mol Genet 2003; 12:1171
-
Jeannet PY, Bassez G, Eymard B, et al. Clinical and histologic findings in autosomal centronuclear myopathy. Neurology 2004; 62:1484.
-
Jeannet PY, Bassez G, Eymard B, et al. Clinical and histologic findings in autosomal centronuclear myopathy. Neurology 2004; 62:1484
-
Zhang J, Ding L, Holmfeldt L, Wu G, Heatley SL, Payne-Turner D, Easton J, Chen X, Wang J, Rusch M, Lu C, Chen SC, Wei L, Collins-Underwood JR, Ma J, Roberts KG, Pounds SB, Ulyanov A, Becksfort J, Gupta P, Huether R, Kriwacki RW, Parker M, McGoldrick DJ, Zhao D, Alford D, Espy S, Bobba KC, Song G, Pei D, Cheng C, Roberts S, Barbato MI, Campana D, Coustan-Smith E, Shurtleff SA, Raimondi SC, Kleppe M, Cools J, Shimano KA, Hermiston ML, Doulatov S, Eppert K, Laurenti E, Notta F, Dick JE, Basso G, Hunger SP, Loh ML, Devidas M, Wood B, Winter S, Dunsmore KP, Fulton RS, Fulton LL, Hong X, Harris CC, Dooling DJ, Ochoa K, Johnson KJ, Obenauer JC, Evans WE, Pui CH, Naeve CW, Ley TJ, Mardis ER, Wilson RK, Downing JR, Mullighan CG (2012). "The genetic basis of early T-cell precursor acute lymphoblastic leukaemia". Nature 481 (7380): 157–63. doi:10.1038/nature10725. PMC 3267575. PMID 22237106
-
Jungbluth,, H. "Pathogenic mechanisms in centronuclear myopathies.". Front Aging Neurosci. doi:10.3389/fnagi.2014.00339. PMID 25566070
-
Bitoun M, Maugenre S, Jeannet PY, et al. Mutations in dynamin 2 cause dominant centronuclear myopathy. Nat Genet 2005;37:1207–9
-
Zuchner S, Noureddine M, Kennerson M, et al. Mutations in the pleckstrin homology domain of dynamin 2 cause dominant intermediate Charcot-Marie-Tooth disease. Nat Genet 2005;37:289–94

 Meet our Residency Program Director
Meet our Residency Program Director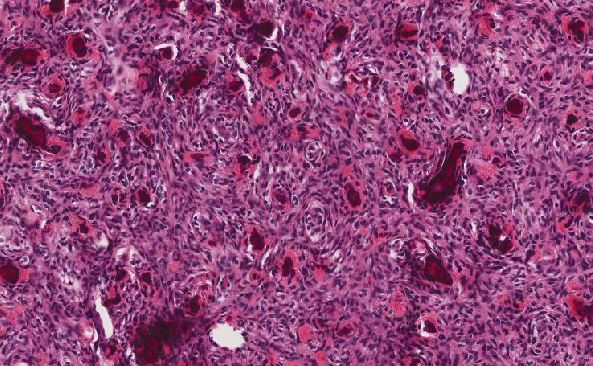
 LeShelle May
LeShelle May Chancellor Gary May
Chancellor Gary May