Residency Program - Case of the Month
April 2012 - Presented by Christina Di Loreto, M.D.
Answer:
Adenoma malignum (minimal deviation adenocarcinoma)
Microscopic description:
On microscopic examination, the tumor consists of glands of variable size that range from small to regular to large with a periglandular stromal response in some areas. Many of the glands have bizarre, complex contours. Glands are also found adjacent to thick-walled blood vessels. The glands are lined by a single layer of cuboidal to columnar mucin-producing cells with basally oriented nuclei and inconspicuous nucleoli. Cytologic features range from normal to mildly atypical. Mitotic figures are rare.
Discussion:
Adenoma malignum, also known as minimal deviation adenocarcinoma, is a rare subtype of well-differentiated adenocarcinoma that accounts for approximately 1% of all cervical adenocarcinomas. The age range of patients diagnosed with adenoma malignum is broad (25-72 years), occurring at a mean age of 42 years. Most cases are sporadic, though patients with Peutz-Jeghers syndrome (PJS) are at increased risk. [1, 2, 3, 4], Mutations of the STK11 gene, a tumor suppressor gene responsible for PJS and located on chromosomal band 19p13.3, has been found in this tumor type. [4, 5, 6] Prediction of adenoma malignum is a diagnostic challenge by cytology, and, unlike other endocervical adenocarcinomas, high-risk human papillomavirus (HPV) infection and p53 gene alterations have only been found in occasional tumors. [2, 4, 7] Synchronous or metachronous ovarian mucinous neoplasms with histologic features resembling cystadenoma, borderline tumor, or well-differentiated adenocarcinoma may also occur and be confused with metastases or a primary ovarian tumor. [1, 2, 3]
As with most cervical carcinomas, the commonest presenting symptom is vaginal bleeding. Mucoid or watery vaginal discharge may also be the presenting complaint. [1, 2, 3]
Grossly, the cervix may appear normal, diffusely enlarged and barrel-shaped, polypoid or ulcerative with a firm or indurated cervical wall. Sectioning of the cervix shows thickening by tan-white or yellow tumor pervading the thickness of the endocervical wall. Mucin-filled cysts may be prominent. [1, 2, 3]
Microscopically, the neoplastic glands appear deceptively innocuous; however, they are usually deeply invasive, exemplified by their presence adjacent to thick-walled blood vessels. The glands demonstrate a range of sizes and shapes and can be convoluted with papillary projections into the lumina. Mucin-rich columnar (and occasionally cuboidal) epithelial cells with basal nuclei and inconspicuous nucleoli line the glands. The cytologic features may be normal, mildly atypical, or, in small foci, overtly malignant. Mitoses are rare. A stromal response may be present around neoplastic glands, though this finding is often focal. [1, 2, 3] Vascular and perineural invasion is often present. [2, 4] Deep invasion and transmural spread or spread to the parametrium is frequently seen, and metastasis to lymph nodes is common. [1, 2]
The differential diagnosis for adenoma malignum includes many benign lesions, such as diffuse laminar endocervical glandular hyperplasia, deep nabothian cysts, adenomyoma of endocervical type, endocervical glandular tunnel clusters, lobular endocervical glandular hyperplasia, and endocervicosis. Special stains and immunohistochemistry may be useful for distinguishing adenoma malignum from these other benign processes. A combined Alcian blue (pH 2.5)-periodic acid Schiff (PAS) stain will stain glands of adenoma malignum red due to the presence of neutral mucin; normal endocervical glands will stain purple because of the high content of acid and neutral mucins. Reticulin staining will demonstrate basement membrane disruption in malignant glands. Extensive cytoplasmic staining with carcinoembryonic antigen (CEA) and an elevated Ki-67 also suggest malignancy. Positivity for alpha-smooth muscle actin and decreased or absent estrogen receptor (ER) positivity in stromal cells is also seen around malignant glands. [1,2] It has also been shown that neoplastic glands of adenoma malignum positively stain with HIK1083, an antibody for pyloric gland mucin. [2, 3]
The literature concerning the prognosis of adenoma malignum is conflicting. Gilks et al found that 50% of patients with stage 1 tumors and 80% of patients with stage 2 tumors died of recurrent disease despite radical treatment in a majority of cases in a literature review of pretreatment clinically staged tumors [1, 7] It has been argued that these results are due to clinical understaging and undertreatment and that the prognosis is actually consistent with other forms of well-differentiated adenocarcinomas; Lim et al report that 16 of 18 cases of adenoma malignum in their study showed no clinical evidence of recurrence after a mean follow-up period of 49.2 months with a 5-year survival rate of 88.8%. [8]
References:
- Robboy SJ, Mutter GL, Prat J, Bentley RC, Robboy’s Pathology of the Female Reproductive Tract. Second Edition. London: Churchill Livingstone, 2009.
- Clement PB and Young RH. Atlas of Gynecologic Surgical Pathology. Second Edition. Philadelphia: Elsevier, 2008.
- Mills SE, Carter D, Greenson JK, Reuter VE, and Stoler MH. Sternberg’s Diagnostic Surgical Pathology. Philadephia: Lippincott Williams & Wilkins, 2010.
- Rosai J. Rosai and Ackerman’s Surgical Pathology. Tenth Edition. London: Mosby, 2011.
- Kuragaki C, et al. Mutations in the STK11 gene characterize minimal deviation adenocarcinoma of the uterine cervix. Lab Invest 2003;83:35-45
- Lee JY, et al. A distinct region of chromosome 19p13.3 associated with the sporadic form of adenoma malignum of the uterine cervix. Cancer Res 1998;58:1140-1143.
- Toki T, Zhai YL, Park JS, Fujii S. Infrequent occurrence of high-risk human papillomavirus and of p53 mutation in minimal deviation adenocarcinoma of the cervix. Int J Gynecol Pathol 1999;18:215-219.
- Gilks CB, Young RH, Aguirre P, DeLellis RA, Scully RE. Adenoma malignum (minimal deviation adenocarcinoma) of the uterine cervix: a clinicopathological and immunohistochemical analysis of 26 cases. Am J Surg Pathol 1989;13:717-729.
- Lim K-T, et al. Adenoma malignum of the uterine cervix: clinicopathologic analysis of 18 cases. Kaohsium J Med Sci 2012;28:161-164.

 Meet our Residency Program Director
Meet our Residency Program Director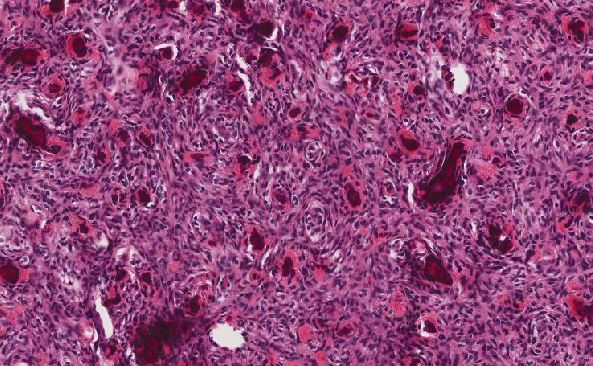
 LeShelle May
LeShelle May Chancellor Gary May
Chancellor Gary May