Residency Program - Case of the Month
January 2011 - Presented by Mahan Matin, M.D.
Answer:
Low grade Mullerian adenosarcoma
Histologic description
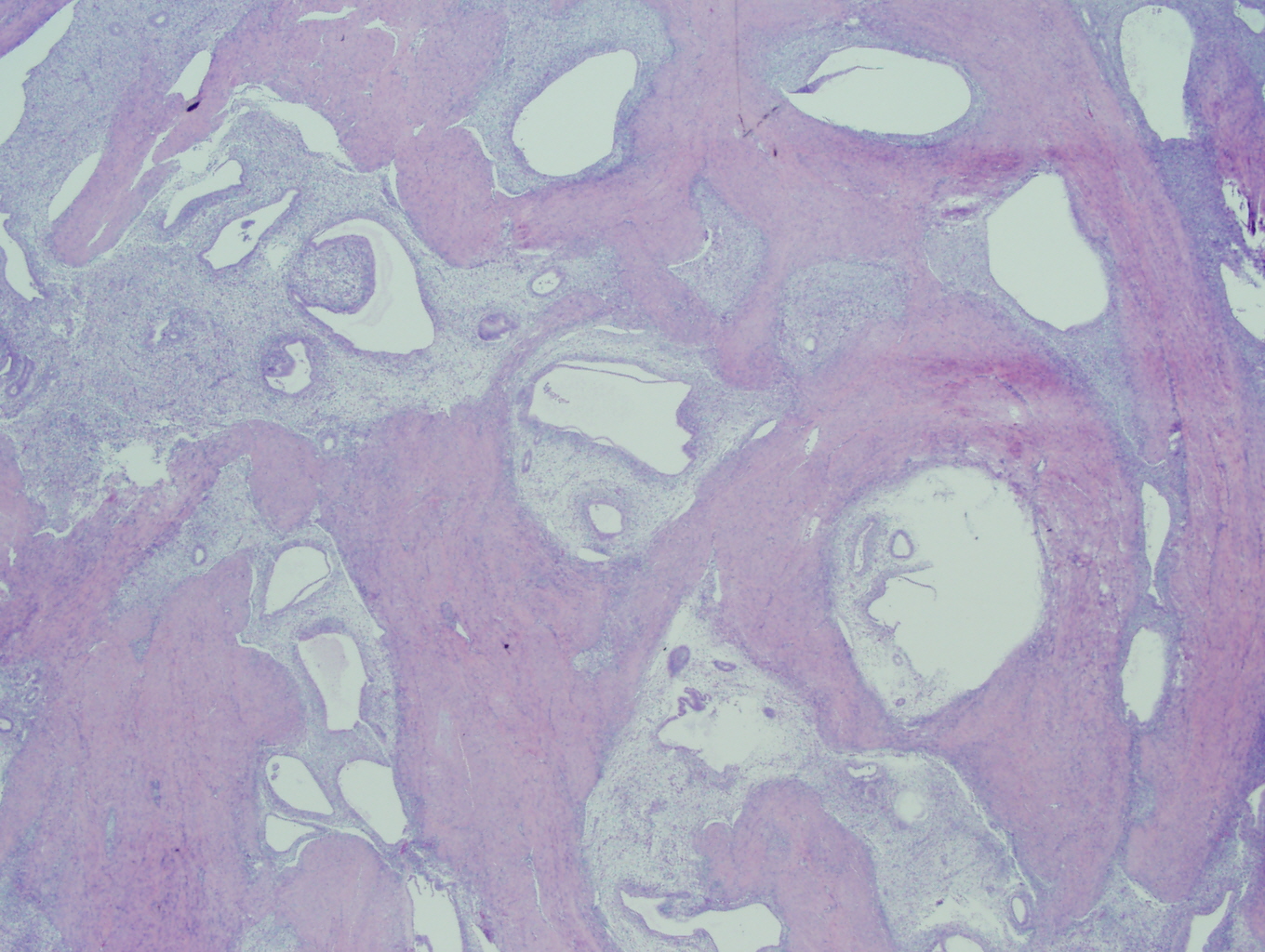
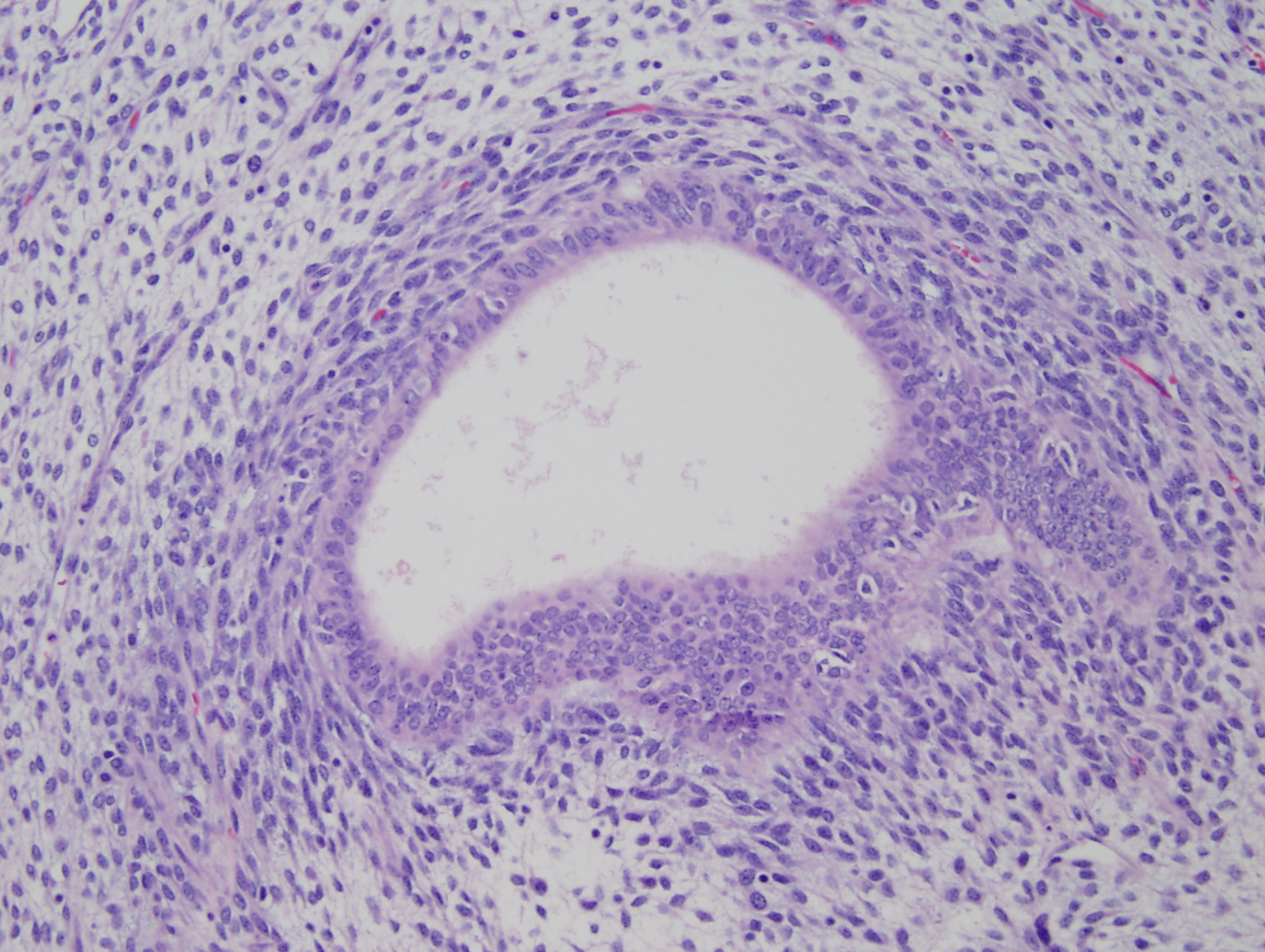
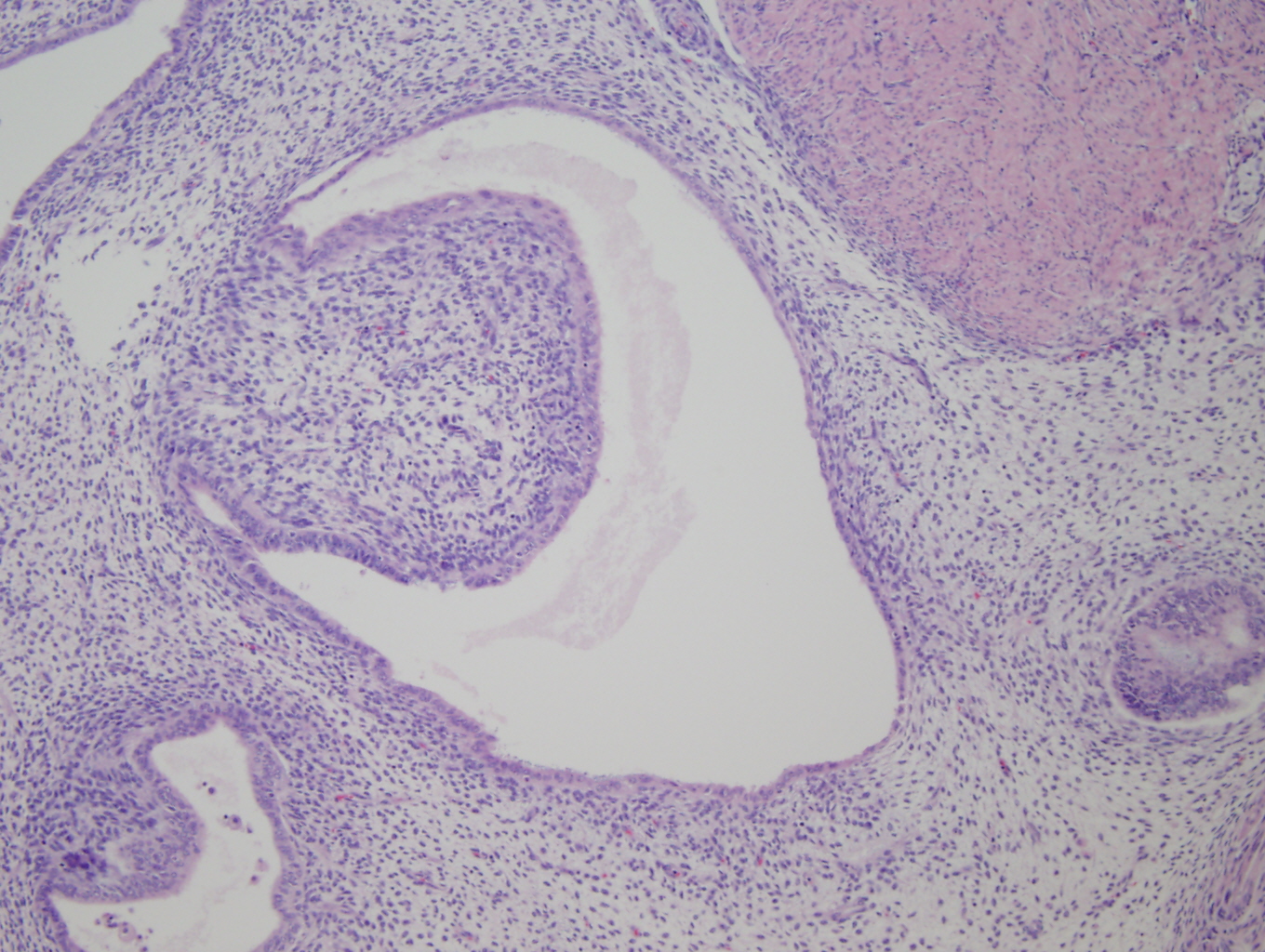
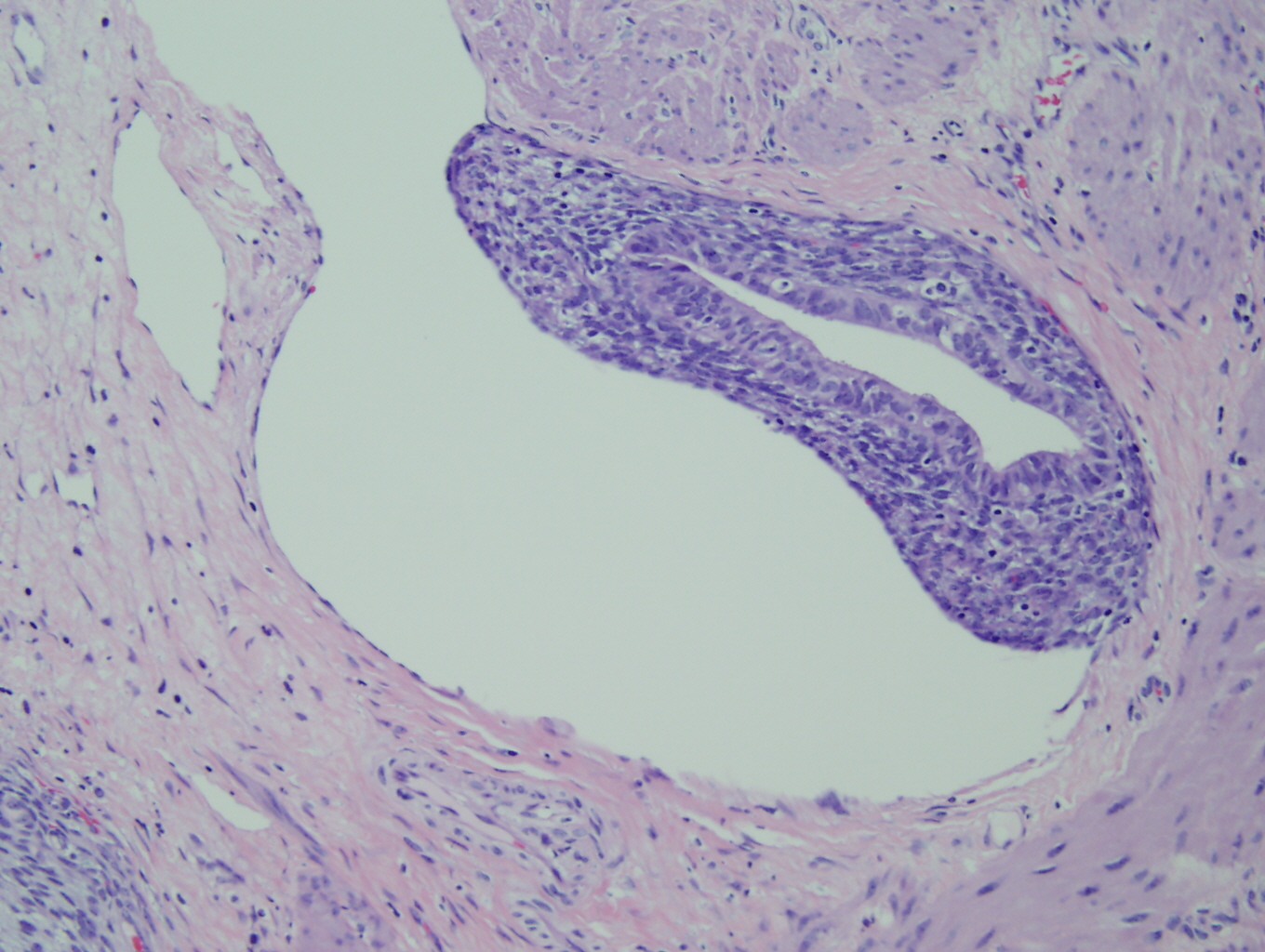
Histologic sections of the myometrium demonstrate a deeply and extensively infiltrating biphasic proliferation of spindled cells admixed with endometrial-type glands characteristic of endometrial tissue (Figure 1). There are benign glands lined by cuboidal epithelium, surrounded by a cellular stroma having moderate atypia. Periglandular cuffing or stromal condensation (Figure 2), intraglandular polypoid projections (Figure 3) and lymphovascular invasion (Figure 4) are seen.
|
Figure 1: Bland epithelium and dense stromal cellularity |
Figure 2: Periglandular cuffs |
|
Figure 3: Intraglandular polypoid projection |
Figure 4: Lymphovascular invasion |
|
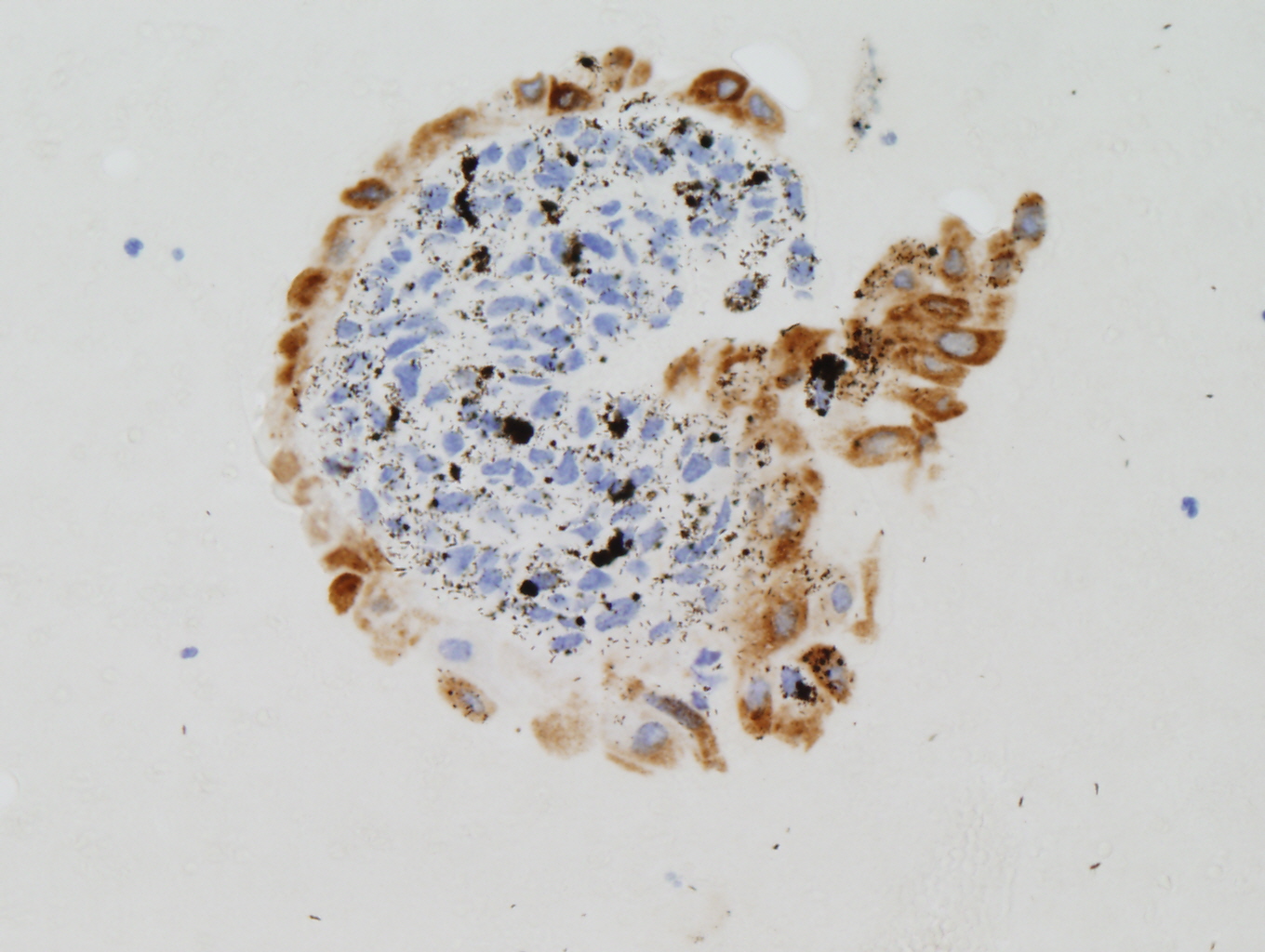
Figure 5: HCG positive |
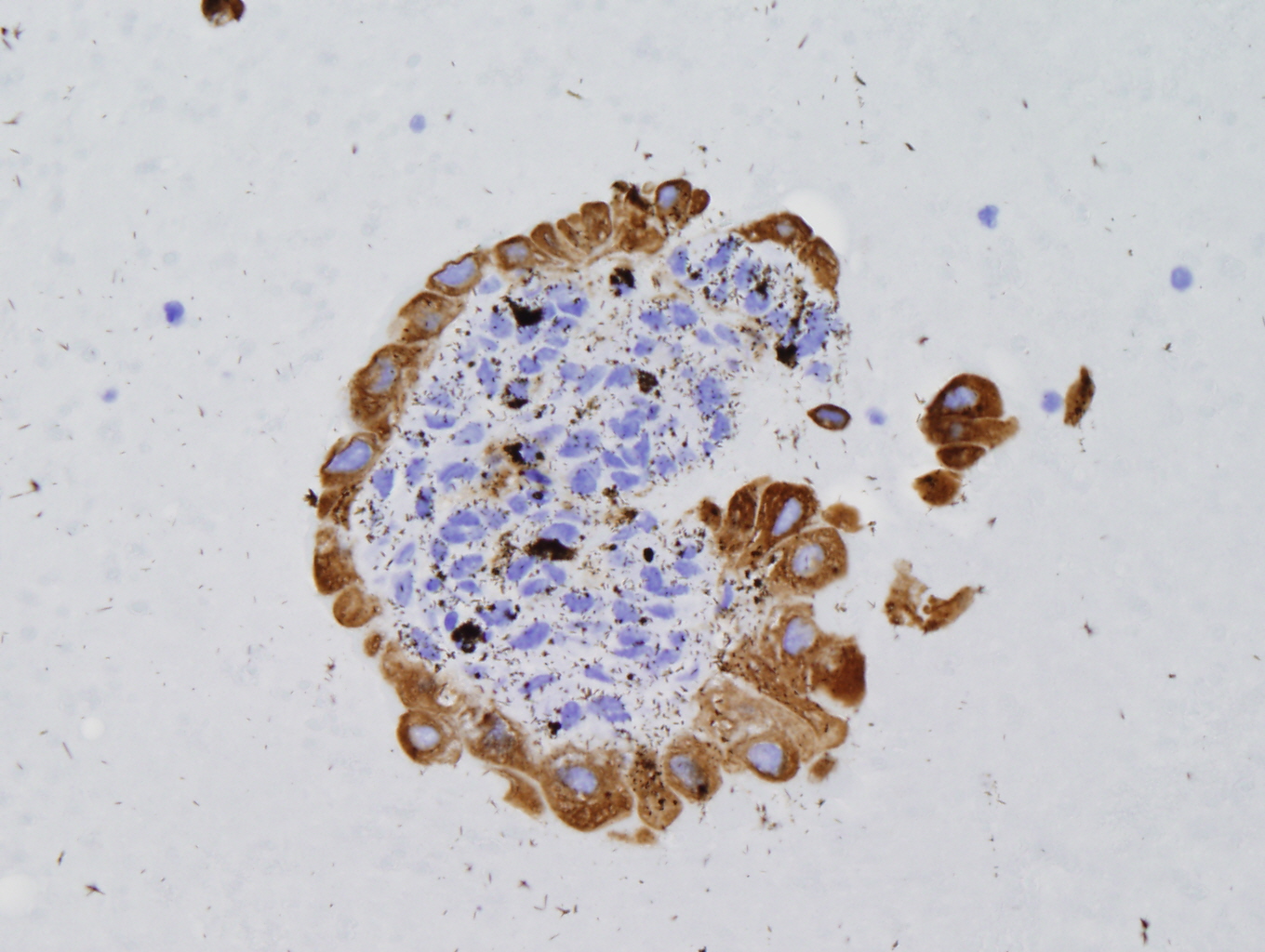
Figure 6: CD10 positive |
|
Figure 7: AE1/AE3 positive |
|
Discussion:
Mullerian adenosarcoma of the uterus is a rare biphasic malignancy along the spectrum of mixed mullerian tumors, composed of benign glandular epithelium in combination with a malignant stromal component (1, 3-4, 5, 8).
| Epithelioid component | Mesenchymal component | Tumor |
| Benign | Benign | Adenofibroma |
| Benign | Malignant | Adenosarcoma |
| Malignant | Benign | Carcinofibroma |
| Malignant | Malignant | Carcinosarcoma |
The tumor is usually found in postmenopausal women and young adults (1, 5), but can occur in women of all ages (4, 8) and in sites other than the uterus, especially the ovary (6). It is a low-grade malignancy associated with local recurrence in up to 30-40% (1, 3). The mortality rate is 20-25% (3, 8). Adenosarcoma in younger patients is often misdiagnosed and more aggressive, and can be extrauterine (2, 4). Distant metastases are extremely rare (1). Some studies show a relationship between the use of tamoxifen and adenosarcoma (4, 7, 8).
Typical signs and symptoms include abnormal vaginal bleeding (most common and especially in young patients), lower abdominal pain, uterine mass or cervical neoplasm (1, 2, 4, 7, 8). Physical exam shows an enlarged uterus and a bulky polypoid mass protruding through the cervical os (7, 8). At the time of diagnosis, most patients have stage I tumors (4).
The tumor is typically seen as a polypoid or sessile endometrial neoplasm with a smooth or knobby surface, and can enlarge the uterus (4, 5). It occurs in the cervix in 5-10% (4). The cut surface can be tan, brown or gray, with necrotic and hemorrhagic areas. Most tumors have small cysts (4, 8).
The hallmark of adenosarcoma is benign glandular epithelium with a malignant mesenchymal component (8). Usually the stroma concentrates around cystic glands and forms periglandular cuffs (5). The glands are typically lined by bland cuboidal or columnar cells (2, 4, 8). The glandular epithelium can be hyperplastic or have focal nuclear atypia, but cannot be malignant (2, 8). The epithelium mostly resembles inactive or proliferative endometrial epithelium, but secretory, hyperplastic, atypical hyperplastic endometrioid, mucinuous, squamous and clear cell types can be seen (4). Most tumors show mild to moderate nuclear atypia in the stroma (4, 5). The mesenchymal component is usually homologous sarcoma, but heterologous mesenchymal elements can be found in 10-15% (1, 2, 4, 5, 8). The mesenchymal component is the probable cause of the symptoms (6).
Myometrial invasion >30% of the uterine wall (seen in ¼ of adenosarcomas), a high-grade sarcomatous component, and stromal overgrowth are associated with a high rate of recurrence or metastasis, which are usually found in the vagina or pelvis (1, 4-6, 8). Tumors with lymphovascular invasion also have an unfavorable outcome (4).
Diagnosis is made based on a combination of "periglandular cuffs" and "intraglandular projections" of sarcomatous stromal cells. The stromal components can have an average of only 1-3 mitotic figures per 10 HPF (4). Immunohistochemistry staining may be helpful (1, 2, 5). Several studies show estrogen and/or progesterone receptors in both the stromal and epithelial components of adenosarcoma (7). In tumors without sarcomatous overgrowth, the immunophenotype of the stromal compartment is similar to that of endometrial stromal sarcoma. With sarcomatous overgrowth, they express higher Ki-67 and p53 and lose ER, PR and CD10 (9). The differential diagnosis includes atypical endocervical polyp, adenomyoma, adenofibroma, carcinosarcoma and embryonal rhabdomyosarcoma (1, 2, 6).
Treatment includes abdominal washings, total abdominal hysterectomy and bilateral salpingooopherectomy (2, 4, 7, 8). Pelvic radiation might be recommended but has little or no impact on survival (3, 7). According to Shi et al, the decision for postoperative chemotherapy can be made based on muscular invasion and the degree of sarcomatous stromal overgrowth (2). Prolonged clinical observation is necessary since recurrence often occurs years later (1, 4).
References:
- Bagga et al, Adenosarcoma of the uterine cervix with heterologous elements: a case report and review of literature: Arch Gynecol Obstet. 2010 Apr;281(4):669-75. Epub 2009 Aug 15.
- Shi et al, The diagnosis and treatment of Mullerian adenosarcoma of the uterus. Aust N Z J Obstet Gynaecol. 2008 Dec;48(6):596-600.
- Arend et al, Long-term outcome and natural history of uterine adenosarcomas. Gynecol Oncol. 2010 Nov;119(2):305-8. Epub 2010 Aug 4.
- Robert J. Kurman, Blaustein's Pathology of the Female Genital Tract, 1994
- Gallardo et al, Mullerian adenosarcoma: a clinicopathologic and immunohistochemical study of 55 cases challenging the existence of adenofibroma. Am J Surg Pathol. 2009 Feb;33(2):278-88.
- Soslow et al, Mullerian adenosarcomas: an immunophenotypic analysis of 35 cases. Am J Surg Pathol. 2008 Jul;32(7):1013-21.
- Tjalma et al, Mullerian adenosarcoma of the uterus associated with long-term oral contraceptive use. Eur J Obstet Gynecol Reprod Biol. 2005 Apr 1;119(2):253-4.
- Robboy et al, Robboy's Pathology of the Female Reproductive Tract, 2 edition (October 29, 2008)
- D'Angelo et al, Uterine sarcomas: a review, Gynecol Oncol. 2010 Jan;116(1):131-9. Epub 2009 Oct 23. Review.


 Meet our Residency Program Director
Meet our Residency Program Director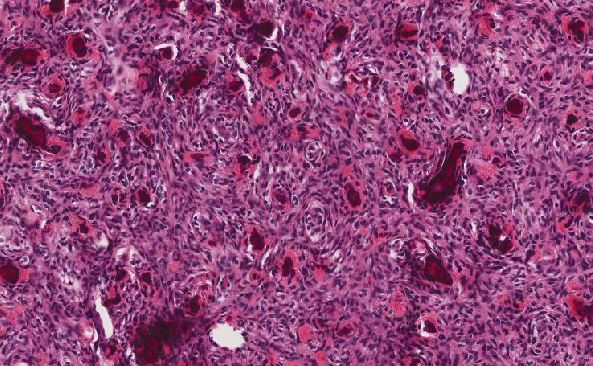
 LeShelle May
LeShelle May Chancellor Gary May
Chancellor Gary May