Residency Program - Case of the Month
April 2010 Final Diagnosis - Presented by Aram Millstein, M.D.
Answer:
Metastatic Melanoma
Histologic description:
The tumor is composed of sheets of epithelioid and spindle cells in the submucosa with little mucosal invasion. Scattered bizarre cells are also present. There is marked nuclear pleomorphism and prominent nucleoli are present. Some cells have multiple nucleoli. There are frequent mitoses, including some atypical forms. There are also scattered cells that contain large granules of brown pigment in their cytoplasm. No necrosis is present. The neoplastic cells are positive for S-100, Melan A, HMB45, c-KIT, and vimentin and negative for CD34, smooth muscle and muscle specific actins, desmin, and Cam 5.2.
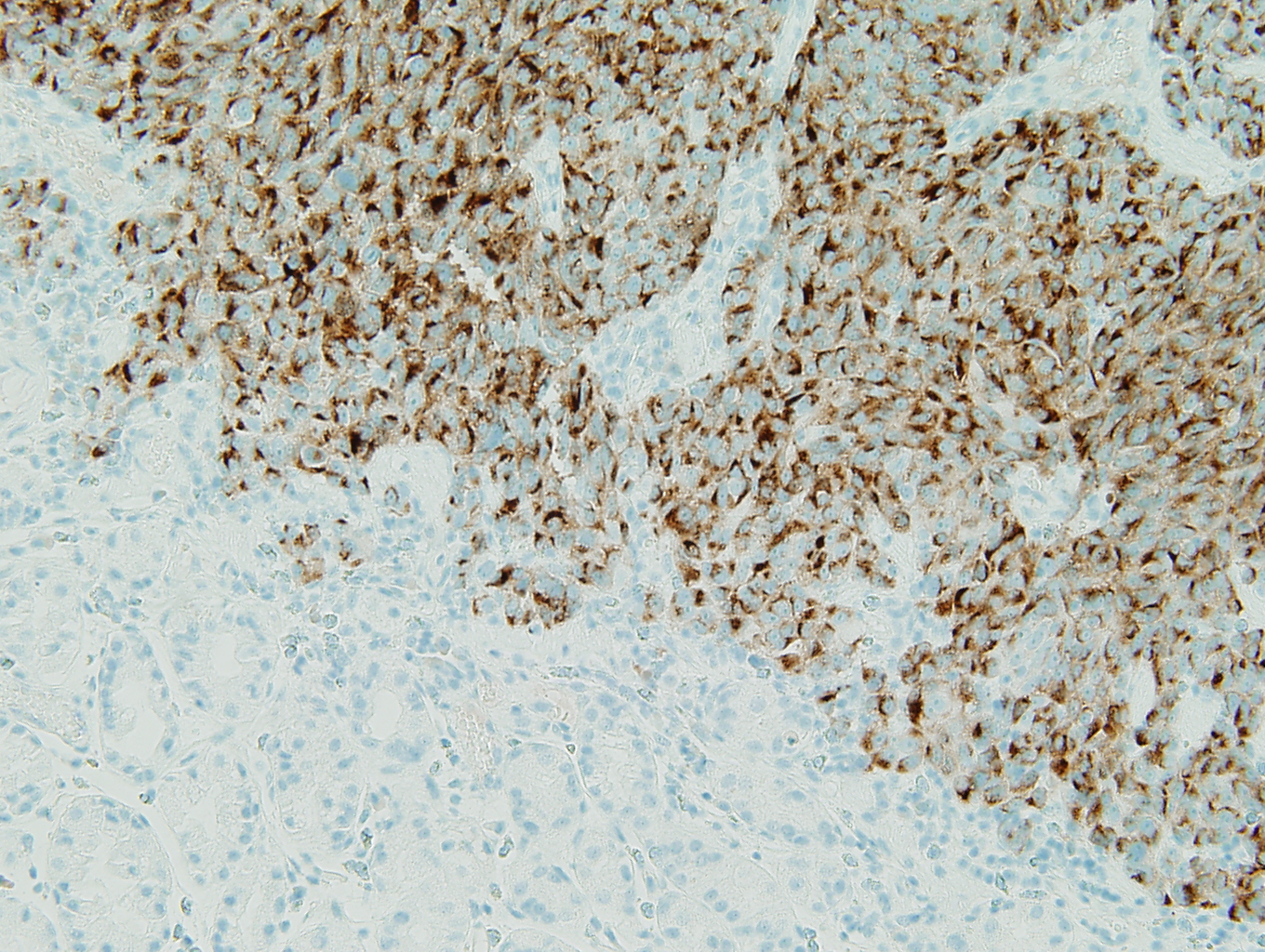
Immunohistochemistry
|
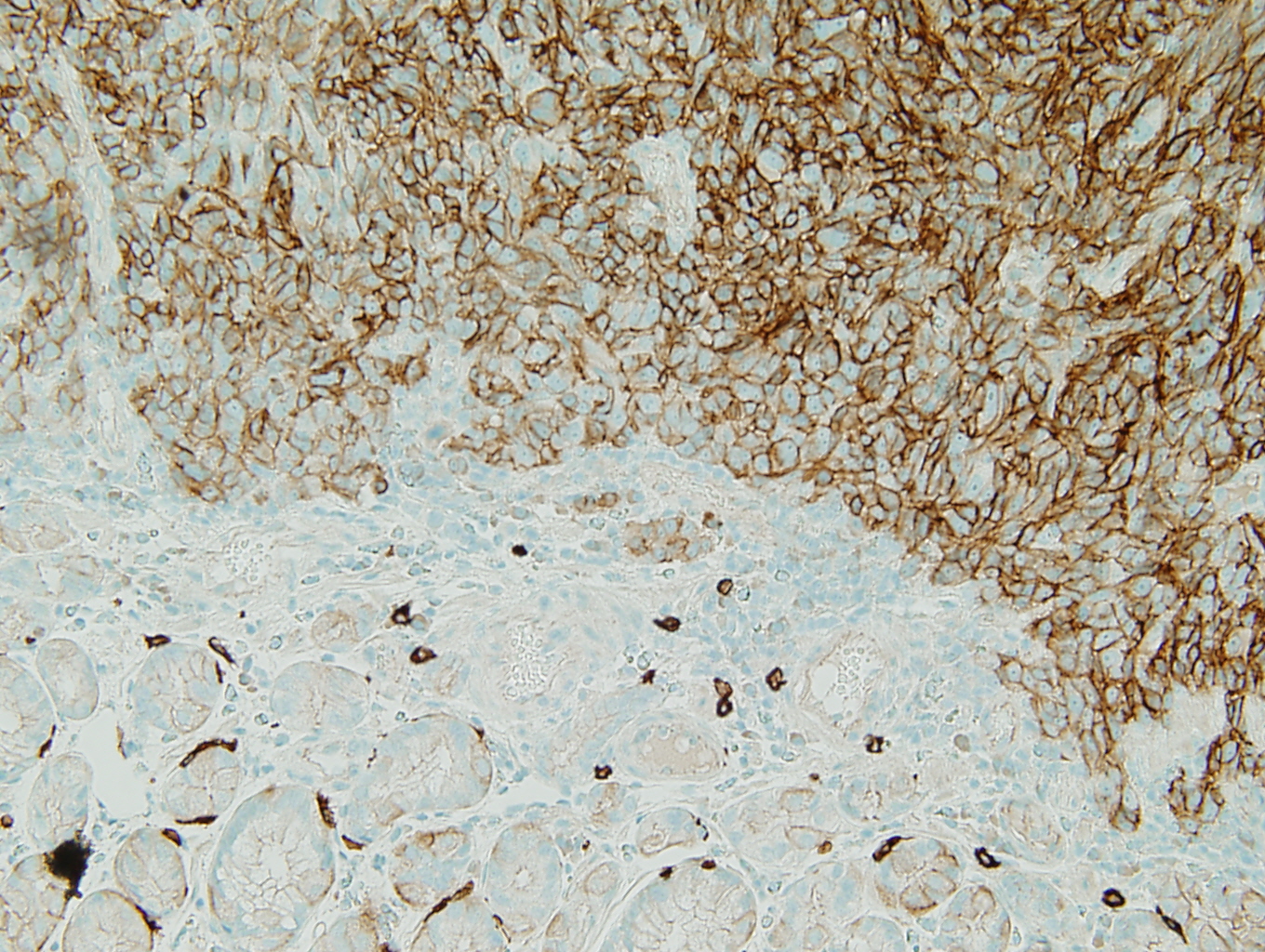
Figure 1: c-KIT |
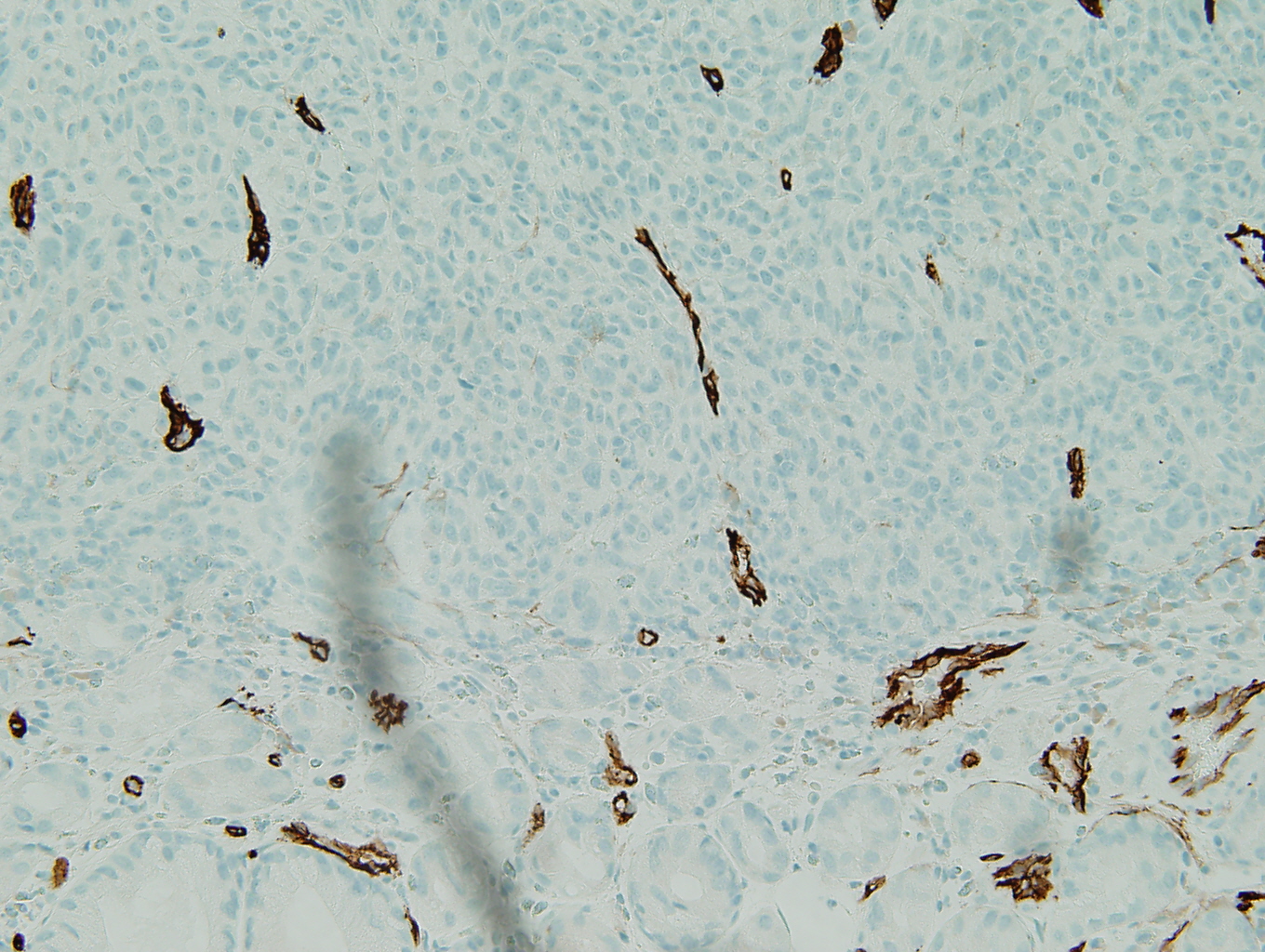
Figure 2: CD34 |
|
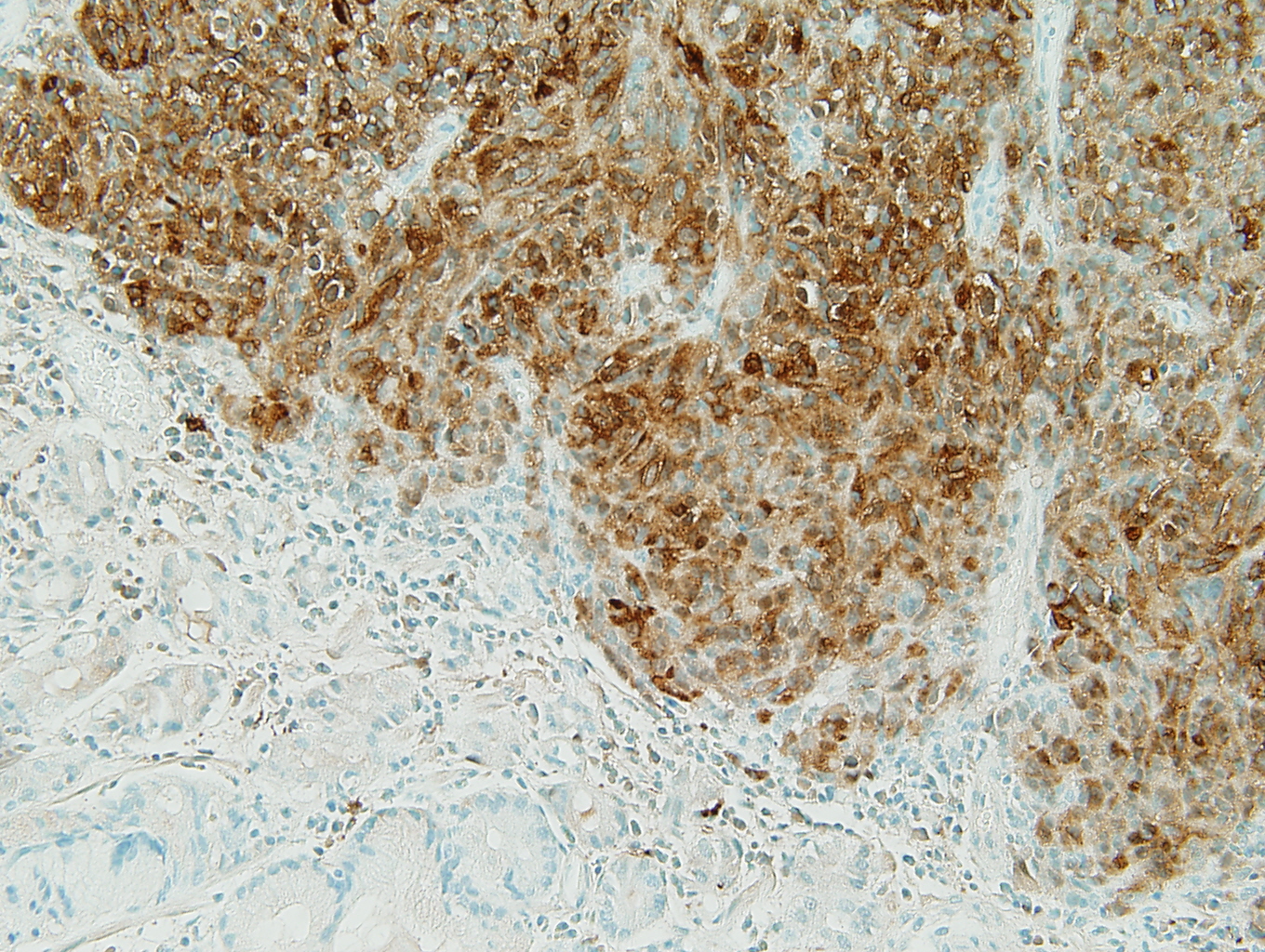
Figure 3: S-100 |
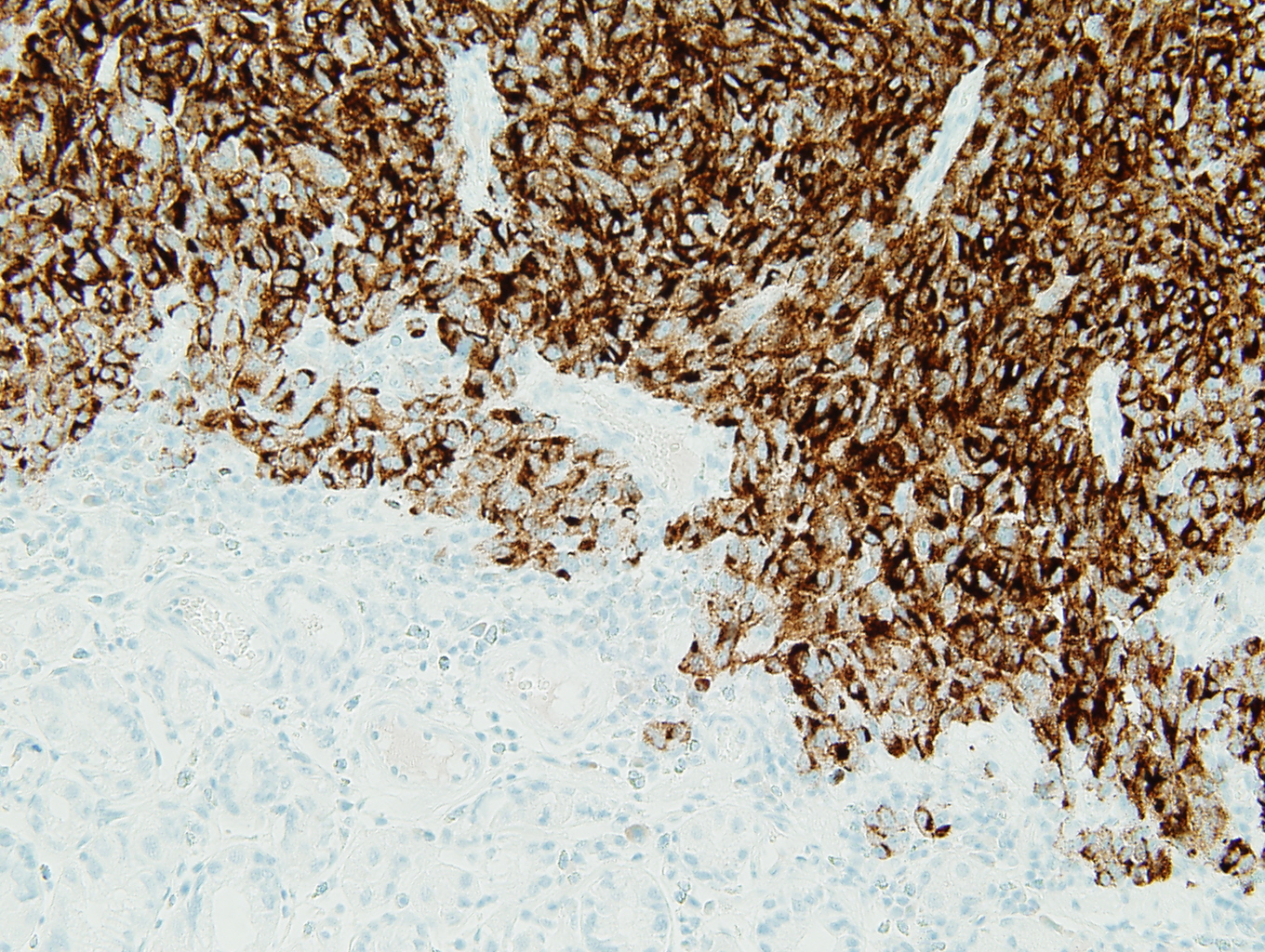
Figure 4: HMB45 |
|
Figure 5: Melan A |
|
Discussion:
The positive S-100, HMB45, and Melan A stains prove the tumor to be a metastatic melanoma. The scattered brown pigment throughout the tumor is also consistent with melanoma. The primary differential is a gastrointestinal stromal tumor (GIST). Discounting the pigment, the tumor has many of the same histologic features of a GIST. GISTs can be mixed spindle cell type and epithelial type cells. A sarcomatous subtype has significant mitotic activity and pleomorphism which is present in this tumor. Ninety-five percent of GISTs are c-kit (CD117) positive (3) and this tumor is strongly positive for c-kit. Ninety-five percent are negative for desmin and 82% are negative for smooth muscle actin (1). Similarly, this tumor is negative for these markers (not shown). The remainder of the immunohistochemistry done provides evidence against identifying the tumor as a GIST. Eighty to 85% of gastric GISTs are positive for CD34 (1); however, this tumor is negative for CD34. GISTs are rarely positive for S-100 (less than 1% [1]), but this tumor is strongly positive for S-100. The fact that this tumor is multifocal (it is two discrete polyps) does not support a diagnosis of a GIST, as GISTs are almost always solitary lesions (3).
Malignant Melanoma does metastasize to the gastrointestinal tract and the time interval between primary melanoma and metastasis can be many years (3) as was the case with this patient. Characteristically, there are multiple metastases to the submucosa with normal overlying mucosa. The cells can be spindle cells or a mixture of spindle and epithelial cells and have large nucleoli. All of which we see in the histology of this tumor. The immunohistochemical profile of this tumor is more consistent with metastatic melanoma. As mentioned S-100, HMB45, and Melan A are positive. CD34 is negative as expected in melanoma (3). CD117 is diffusely positive in this case. Although melanomas generally have weak to focal positive expression of CD117, they can be diffusely positive. Alexis, Martinez, and Lutzky examined CD-117 expression in 8 primary and 32 metastatic melanoma cases in Melanoma Research 2005. Thirty-five of the 40 cases were CD117 positive to some degree and seven of the 40 cases showed moderate to strong staining in greater than 75% of the tumor (2).
This melanoma had areas of epithelioid cells and spindle cells, but some melanomas can consist only of spindle cells. Up to 75% of cases of spindle cell or desmoplastic melanoma can be negative for HMB-45 (3). Although this case was positive for HMB-45, it is important to use multiple markers to evaluate for melanoma. In a study by Lim et al. researchers examined the pattern of melanoma marker expression in 32 cases of desmoplastic melanoma. Specifically, 66% of the cases stained positively for Melan-A, but only 47% of the spindle cells stained positively for Melan-A (4).
A new marker that can assist in difficult cases is microphthalmia transcription factor (MiTF). MiTF is a sensitive marker of epithelioid melanoma. King et al. examined MiTF in 76 cases of melanoma. Of the 76 cases, 19 were melanomas in situ, 50 were conventional melanomas, and 7 were metastatic melanomas. The majority of these cases had an epithelioid cell morphology, but 17 were either primarily spindle cell or spindled/epithelioid. They stained the 76 cases of melanoma for MiTF, HMB-45, and S-100. All of the cases stained positively for MiTF and 91% and 93% stained positively for HMB-45 and S-100 respectively (5). Further studies have attempted to evaluate MiTF in spindle cell melanomas. Koch et al. stained 79 tumors (20 desmoplastic/spindle cell melanomas, 10 scars, 10 neurofibromas, 12 malignant peripheral nerve sheath tumors, 10 atypical fibroxanthomas, 10 clear cell sarcomas, 3 melanotic schwannomas, and 4 cellular blue nevi) for MiTF. The researchers found the sensitivity and specificity of MiTF for desmoplastic/spindle cell melanoma was 55% and 91%, respectively (6).
Finally, this patient did have a history of right ocular melanoma that was treated with surgery 12 years ago. He had one recurrence that was treated with chemotherapy eye drops.
References:
- Miettinen M and Lasota J. Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis and differential diagnosis. Arch Pathol Lab Med 2006; 130:1466-1478.
- Alexis J, Martinez A, and Lutzky J. An immunohistochemical evaluation of c-kit (CD117) expression in malignant melanoma, and results of imatinib mesylate (Gleevec) therapy in three patients. Melanoma Research 2005;15:283-285.
- Mills S. Sternberg's Diagnostic Surgical Pathology Fifth Edition, Wolters Kluwer/Lippincott Williams & Wilkins, 2010
- Lim E, Browning J, MacGregor D, Davis ID, Cebon JS. Desmoplastic melanoma: comparison of expression of differentiation antigens and cancer testis antigens. Melanoma Research 2006; 16(4):347-55.
- King R, Weilbaecher KN, McGill G, Cooley E, Mihm M, Fisher DE. Microphthalmia transcription factor. A sensitive and specific melanocyte marker for MelanomaDiagnosis. Am J Surg Pathol 1999; Sep;155(3):731-738.
- Koch MB, Shih IM, Weiss SW, Folpe AL. Microphthalmia transcription factor and melanoma cell adhesion molecule expression distinguish desmoplastic/spindle cell melanoma from morphologic mimics. Am J Surg Pathol 2001; Jan;25(1):58-64.

 Meet our Residency Program Director
Meet our Residency Program Director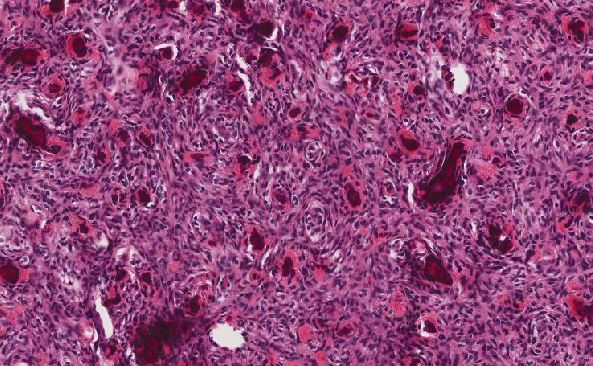
 LeShelle May
LeShelle May Chancellor Gary May
Chancellor Gary May